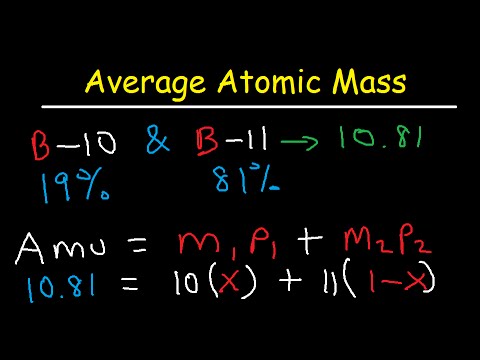
Therefore, this equality holds only for the carbon-12 atom in the stated conditions, and will vary for other substances. However, the mass of an atomic-scale object is affected by the binding energy of the nucleons in its atomic nuclei, as well as the mass and binding energy of the electrons. It follows that the molar mass of a compound (grams per mole) is also numerically close to the average number of nucleons per molecule. The mass in daltons of an atom is numerically very close to the number of nucleons A in its atomic nucleus. However, while this equality can be assumed for almost all practical purposes, it is now only approximate, because of the way the mole was redefined on. A protein whose molecule has an average mass of 64 kDa would have a molar mass of 64 kg/mol. For example, the average mass of one molecule of water is about 18.0153 daltons, and one mole of water is about 18.0153 grams. That is, the molar mass of a chemical compound was meant to be numerically equal to its average molecular mass. The mole is a unit of amount of substance, widely used in chemistry and physics, which was originally defined so that the mass of one mole of a substance, measured in grams, would be numerically equal to the average mass of one of its constituent particles, measured in daltons. The DNA of chromosome 1 in the human genome has about 249 million base pairs, each with an average mass of about 650 Da, or 156 GDa total. Titin, one of the largest known proteins, has an atomic mass of between 3 and 3.7 megadaltons. The molecular masses of proteins, nucleic acids, and other large polymers are often expressed with the units kilodalton (kDa), equal to 1000 daltons, megadalton (MDa), one million daltons, etc. In general, the standard atomic weight of an element is the average weight of its atom as it occurs in nature, expressed in daltons. For example, an atom of helium has a mass of about 4 Da, and a molecule of acetylsalicylic acid (aspirin), C9H8O4, has a mass of about 180.16 Da. This unit is commonly used in physics and chemistry to express the mass of atomic-scale objects, such as atoms, molecules, and elementary particles. As of June 2019, the value recommended by the Committee on Data for Science and Technology (CODATA) is 1.66053906660(50)×10−27 kg, or approximately 1.66 yoctograms. Despite being an official abbreviation for a related obsolete unit and not widely used in the scientific literature, the abbreviation "amu" now often refers to the modern unit (Da or u) in many preparatory texts. It is defined precisely as 1/12 of the mass of an unbound neutral atom of carbon-12 in its nuclear and electronic ground state and at rest. The symbol Da is more widely used in most fields. As of 2019, the International System of Units (SI) lists the dalton, symbol Da, as a unit acceptable for use with the SI unit system and secondarily notes that the dalton (Da) and the unified atomic mass unit (u) are alternative names (and symbols) for the same unit. The current IUPAC endorsed definition is the unified atomic mass unit, denoted by the symbol u. Several definitions of this unit have been used, implying slightly different values. A mass of 1 Da is also referred to as the atomic mass constant and denoted by mu. It is approximately the mass of one nucleon (either a proton or neutron). I know what problem I have is but I do not how I can solve it.The dalton or unified atomic mass unit (SI symbols: Da or u) is a unit of mass widely used in physics and chemistry. No problem is the molecule looks like 'C14-H10-O2' BUT if some of the elements have not one digit after the element, Python reply with this error. # If not, just add that element's mass to the sum. In each iteration, the masses of each element will be added and stored in the mass object Masa = element_mass * int(molecula)# Add the mass of this element by the number of times that element is present. If molecula.isdecimal(): #If the inmediately item is decimal If ch in elements_list: #If the element is in the elements_list,Įlement_mass = masas.get(ch)# Get the mass of this element Molecula = (re.split('(\d )',molecula))#split numbers and elements 
Molecula = re.sub('', '', molecula) #Remove "-" Here is my code: masas = Įlements_list = My code is almost finished BUT I have found a problem I cant solve. (I know, there are very beautiful programs created already but I want to create my own program and feel proud of my self.) I want to design a program to calculate the atomic mass, given a molecule like this 'C14-H10-O'.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
